SO3 Lewis Structure (Sulfur Trioxide) YouTube
To draw this structure, begin by sketching a rough diagram of the molecular arrangement. Next, indicate the lone pairs on each atom and check for any formal charges. If formal charges are present, convert lone pairs to minimize these charges. Repeat this process until all charges are minimized.

SO3 Lewis Structure, Molecular Geometry, and Hybridization Techiescientist
0:00 / 2:55 SO3 2- Lewis Structure - How to Draw the Lewis Structure for SO3 2- (Sulfite Ion) Wayne Breslyn 724K subscribers Join Subscribe Subscribed 1.9K Share 411K views 10 years ago.
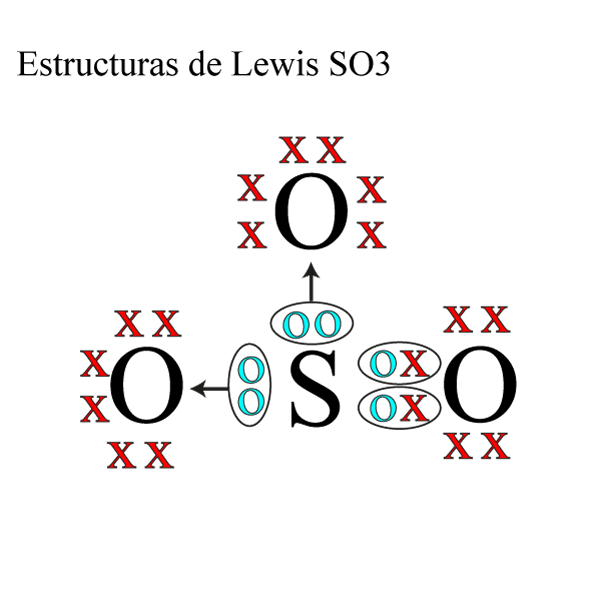
Estructura de Lewis SO3 » Quimica Online
Lewis Structures. Page ID. A Lewis Structure is a very simplified representation of the valence shell electrons in a molecule. It is used to show how the electrons are arranged around individual atoms in a molecule. Electrons are shown as "dots" or for bonding electrons as a line between the two atoms. The goal is to obtain the "best" electron.

Lewis Dot Structure for SO3 (Sulfur trioxide) YouTube
Step 1: Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. When drawing the structure of an ion, be sure to add/subtract electrons to account for the charge. Step 2: Connect the atoms to each other with single bonds to form a "skeleton structure.".

SO3 Lewis StructureLewis structure of SO3 (Sulfur trioxide) YouTube
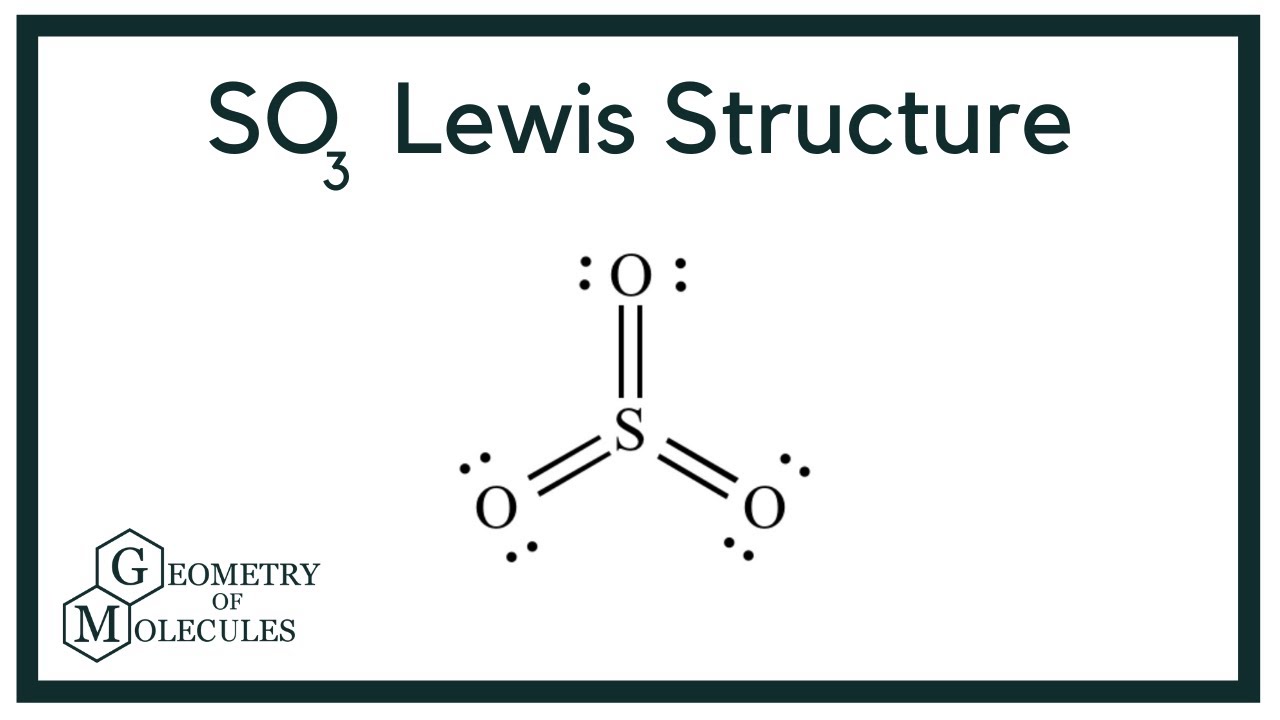
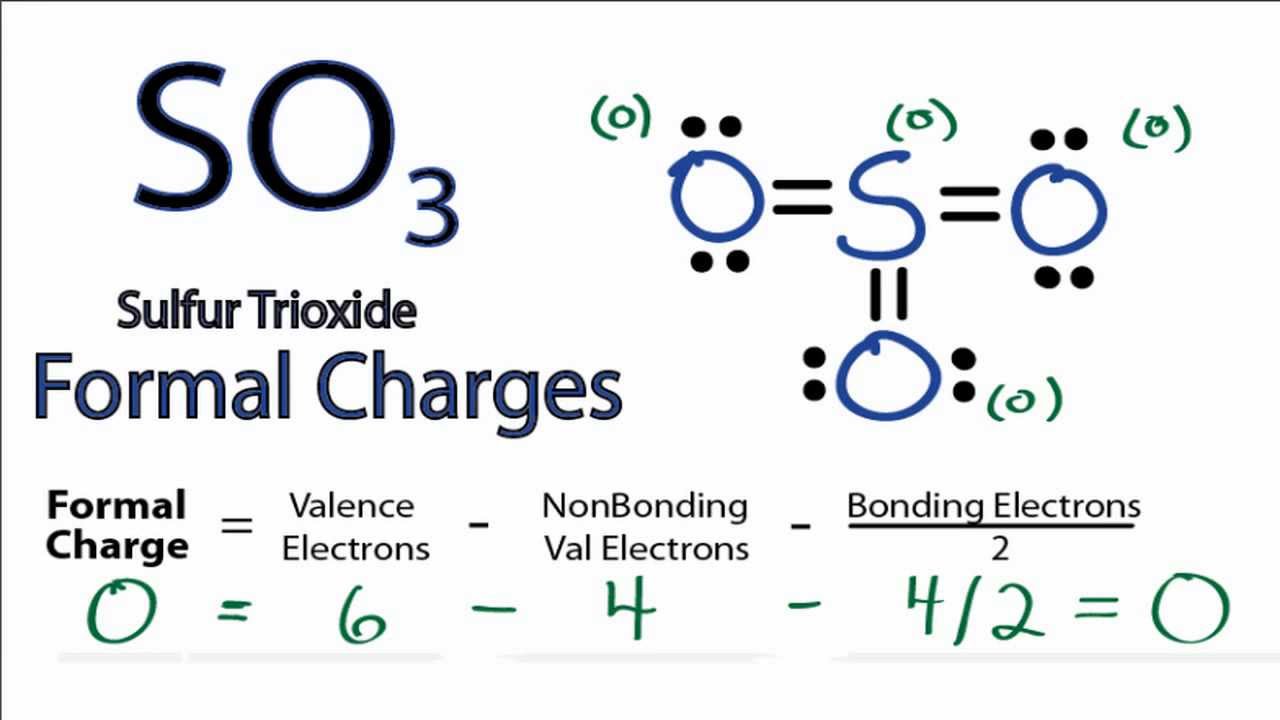
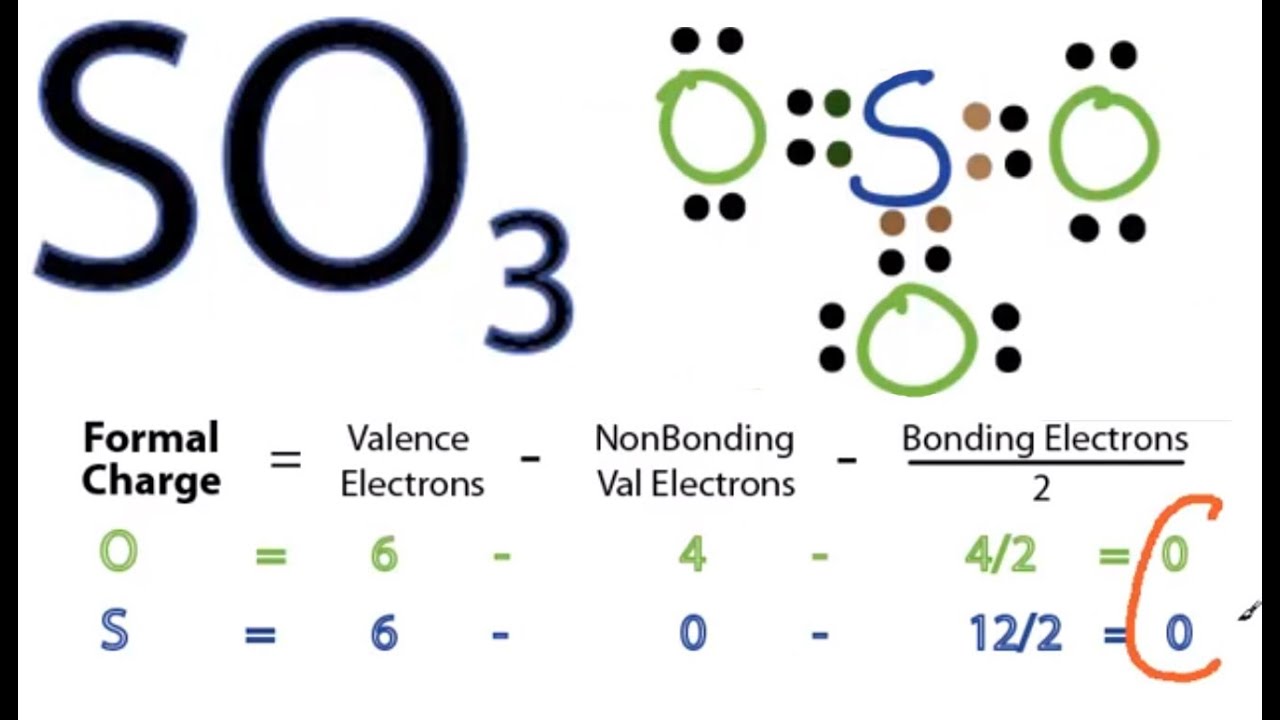
Lewis structure of SO3 (or Sulfur trioxide) contains three double bonds between the Sulfur (S) atom and each Oxygen (O) atom. The Sulfur atom (S) is at the center and it is surrounded by 3 Oxygen atoms (O). The Sulfur atom does not have a lone pair while all the three Oxygen atoms have 2 lone pairs.

Draw the Lewis dot structure for SO3 Brainly.in
Steps of drawing SO3 lewis structure Step 1: Find the total valence electrons in SO3 molecule. In order to find the total valence electrons in SO3 (sulfur trioxide) molecule, first of all you should know the valence electrons present in sulfur atom as well as oxygen atom. (Valence electrons are the electrons that are present in the outermost orbit of any atom.)
Lewis Dot Diagram For So3 Wiring Diagram
Sulfur Trioxide Molecular Geometry Being an intelligent and well-practiced human being, you must know what is molecular geometry, but let me revise it for the all young students out there. Molecular geometry is the three-dimensional structure of the atoms which helps in the constitution of a molecule.
Draw The Lewis Dot Structure For So3 2 slidesharedocs
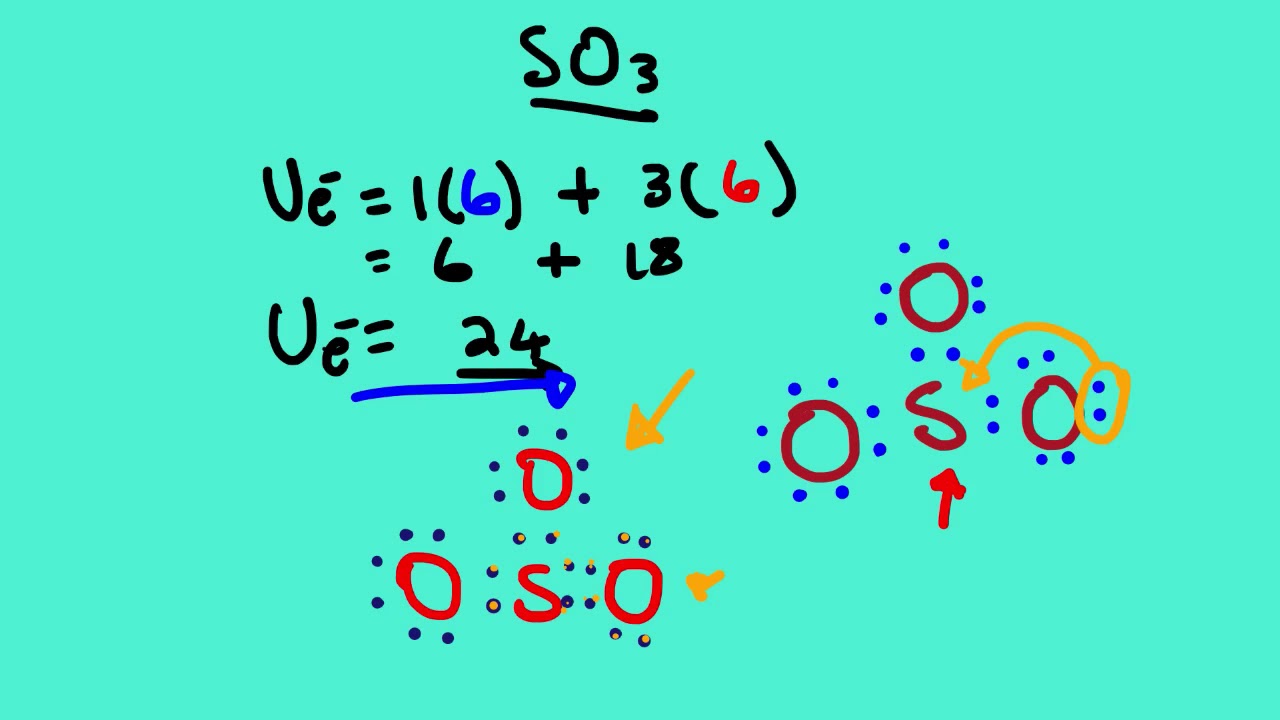
-the physical properties of a molecule such as boiling point, surface tension, etc. Drawing the Lewis Structure for SO 3 ( Sulfur Trioxide) SO 3 is the primary contributer to acid rain in the atomsphere. It is a form of pollution. SO 3 is named Sulfur Trioxide. There are 32 valence electrons available for the Lewis structure for SO 3.

Lewis Dot Structure For So3 slidesharedocs
A step-by-step explanation of how to draw the SO3 Lewis Dot Structure (Sulfur trioxide).For the SO3 structure use the periodic table to find the total number.

make structure of hybridization of SO3 Brainly.in
Let's do the SO3 Lewis structure. Sulfur has 6 valence electrons. Oxygen has 6, but we've got three Oxygens, for a total of; 6 plus 18; 24 valence electrons. Let's put Sulfur at the center and then the Oxygens around the outside, all three of them. Now we'll put two valence electrons between each atom to form a chemical bond.
SO3 Lewis Structure How to Draw the Lewis Structure for SO3 (Sulfur Trioxide) YouTube
The sulfur trioxide is a tetra atomic chemical molecule where both the sulfur and three oxygen molecules bond with an equal number of valence electrons. The diagram is drawn showing dots of valence electrons around the symbol of both sulfur and oxygen atoms with lines predicting bond formation.
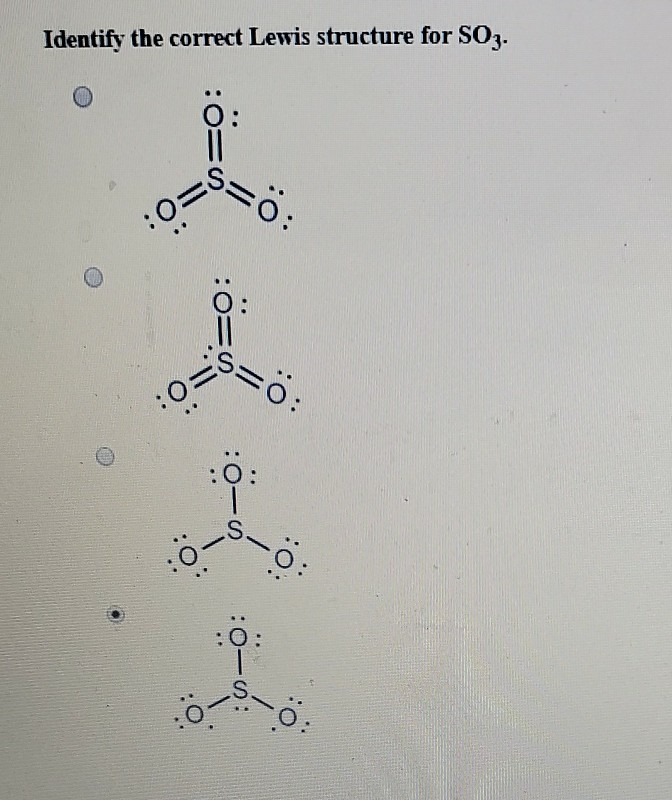
Solved Identify the correct Lewis structure for SO3.
0:00 / 4:53 Lewis Structure of SO3 (Sulfur Trioxide) chemistNATE 259K subscribers Subscribe Subscribed 4.9K Share 625K views 9 years ago Lewis Structures How to draw the Lewis Structure of.

How to draw SO3 Lewis Structure? Science Education and Tutorials
Website-http://www.kentchemistry.com/links/bonding/LewisDotTutorials/SO3.htmI quickly take you through how to draw the Lewis Structure of SO3 (Sulfur Trioxid.
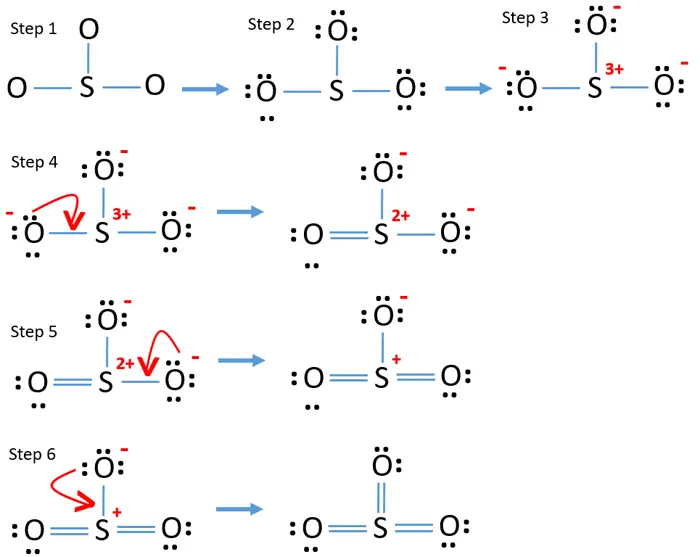
steps of drawing SO3 lewis structure VSEPR method
By Aditi Roy The Lewis structure of SO3, also known as sulfur trioxide, is a representation of how the atom s are arranged in the molecule. It helps us understand the bonding and electron distribution within the compound. In the Lewis structure of SO3, sulfur is the central atom bonded to three oxygen atoms.

Lewis Dot Structure For So3 slidesharedocs
SO3 Lewis Structure - Sulfur Trioxide The Organic Chemistry Tutor 7.24M subscribers Subscribe Subscribed 607 70K views 3 years ago This chemistry video explains how to draw the Lewis.
LEWIS DIAGRAM FOR SO3 YouTube
Sulfur trioxide is a oxide of sulfur and colourless inorganic gas. Also it is a toxic gas. Sulfur trioxide gas is produced due to oxidation of sulfur dioxide gas in air. Lewis structure of SO 3 molecule There are three double bonds around sulfur atom with oxygen atoms in SO molecule. Each oxygen atom has two lone pairs in SO 3 lewis structure.